|
This type of PCR technique uses four primers for DNA amplification (two primers for each strand of the DNA target). The higher temperatures during the initial cycles help primers to bind to DNA templates with greater specificity while the lower temperatures allow more efficient amplification from the produced amplicons. It is achieved by raising the annealing temperature above the melting temperature of the used primers in the initial cycles and lowering in the later cycles. Touchdown PCR is another technique to reduce nonspecific amplification. Non-mechanical hot start PCR uses specialized enzyme systems which inhibit an activation of the DNA polymerase at room temperature. Mechanical hot start PCR performed by heating the reaction mixture to the DNA melting temperature before adding the Taq polymerase. Two variants of this technique are mechanical and non-mechanical hot start PCR. This modification prevents the amplification during reaction setup when primers bind to DNA sequences with low homology. Hot-start PCR technique keeps the DNA polymerase in an inactive state at temperatures lower than an annealing temperature. This type of polymerase chain reaction serves to reduce non-specific amplification during the initial set up stages. The product of this reaction serves as a source of target DNA to a second PCR using the second set of primers. The first set allows a first polymerase chain reaction. This technique utilizes two sets of primers. Nested PCR is used to increase the specificity of a DNA amplification reducing unspecific products. They have to have similar annealing temperatures and produce amplicons of different sizes to form distinct gel electrophoresis bands for the followed PCR analysis. To work properly within one reaction, sets of primers must be optimized. It reduces the consumption of PCR reagents, and, at the same time, imposes restrictions on used primers. 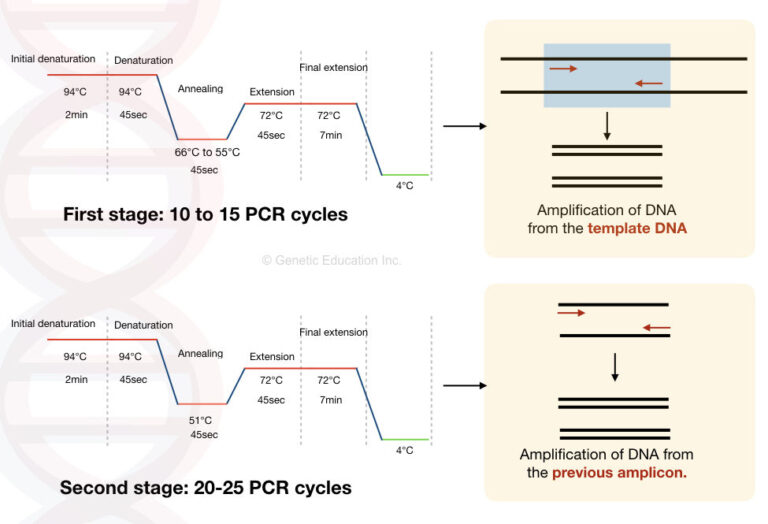
Multiplex PCR is a type of PCR technique which allows an amplification of many target sequences concurrently in the same reaction mixture.Ī single reaction mixture includes sets of primer pairs for different DNA targets. Thermocycling techniques use temperature cycling to drive repeated cycles of DNA synthesis. Nucleic Acids Res. 1990 18(21):6409-6412.Different types of PCR technique based on thermocycling (heating and cooling steps) Optimization of the annealing temperature for DNA amplification in vitro. Understand the importance of melting temperature in molecular biology applications Optimal annealing temperatures give the highest product yield of the correct amplicon. Conversely, when T a is too high reaction efficiency may be reduced because the likelihood of primer annealing is reduced significantly. This can lead to nonspecific PCR amplification and will consequently reduce the yield of the desired product. One consequence of having T a too low is that one or both primers will anneal to sequences other than the intended target, because internal single-base mismatches or partial annealing may be tolerated. The optimal annealing temperature (T a Opt) for a given primer pair on a particular target can be calculated as follows: T a Opt = 0.3 x (T m of primer) + 0.7 x (T m of product) – 14.9, where T m of primer is the melting temperature of the less stable primer-template pair, and T m of product is the melting temperature of the PCR product. Generally, you should use an annealing temperature about 5☌ below the T m of your primers. The annealing temperature (T a) chosen for PCR relies directly on length and composition of the primers. Target Capture Probe Design & Ordering Tool.Library Concentration Conversion Calculator. 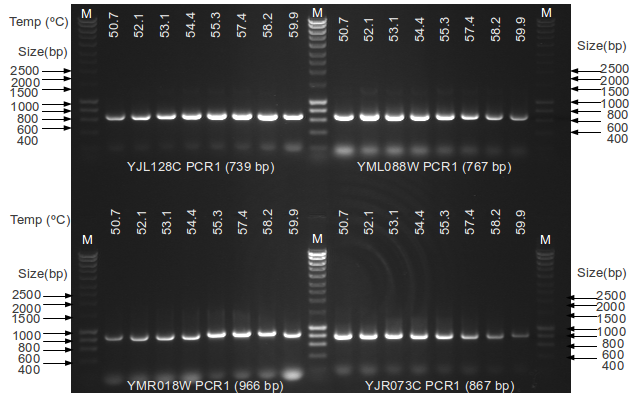
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
